Whitbarrow is that bright white limestone cliff just by the A590 as you head towards Barrow, and a several-mile-long plateau of pavement, crag, woodland and grassland beyond. The group’s visited before- in early 2024- but that was on the western side by Witherslack Hall. This time, we were going to look at the south end, White Scar Quarry.
Now I’ve walked through it plenty of times in previous lives, though never stopped to consider the lichens, or how the quarry was operating. It’s a dramatic place: limestone cliffs tower above the big slab of a quarry floor and, if you’re there at the right time, the Raven and Peregrine calls echo. It was a small and select group who met there last week- just the six of us, and only two to look at lichens. On a hot morning, with clear blue skies, it felt almost like the Cevennes- or somewhere similarly southern. The swathes of yellow flowers on the slopes above caught our eye: was it horseshoe vetch?
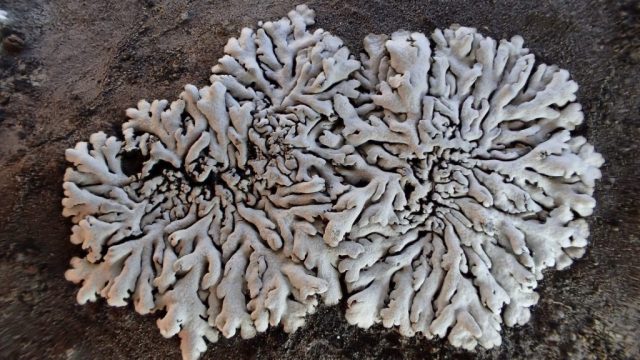
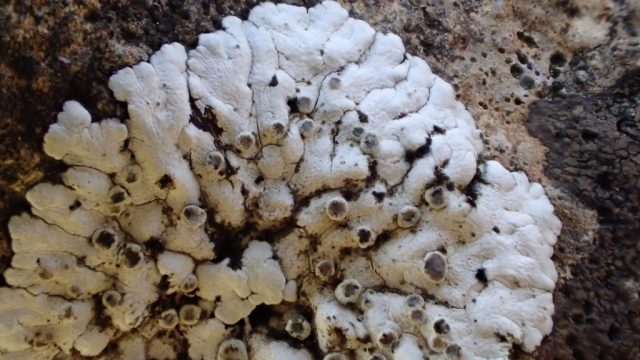
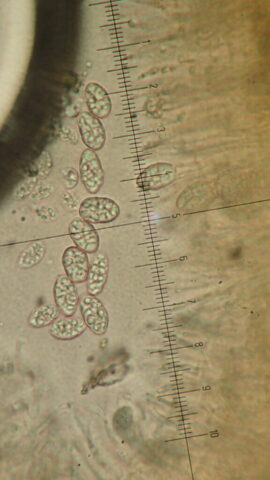
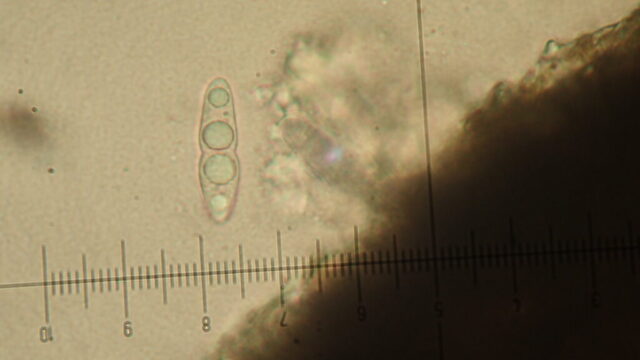
I’m sorry to say that it quickly became apparent that the quarry was a disappointment on the lichen front. The cliffs, slab floor and boulders that remained just didn’t have many lichens. Ok, we found some of the standard limestone stuff: Verrucaria nigrescens; Lathagrium fuscovirens and L. auriforme; Variospora flavescens and Scytinium pulvinatum; Protoblastenia calva and P. rupestris. But there wasn’t that much of anything, and the rock cracks which are often interesting only once came up with Placidium squamulosum. A bright spot was the thalli of Leproplaca cirrochroa, which has questing orange lobes and lovely bright yellow soredia. There’s fewer than 50 records for Cumbria. These are mostly in the south- I guess cos it likes sunny limestone. But we didn’t see any Circinaria calcarea until well after lunch, which tells a story.
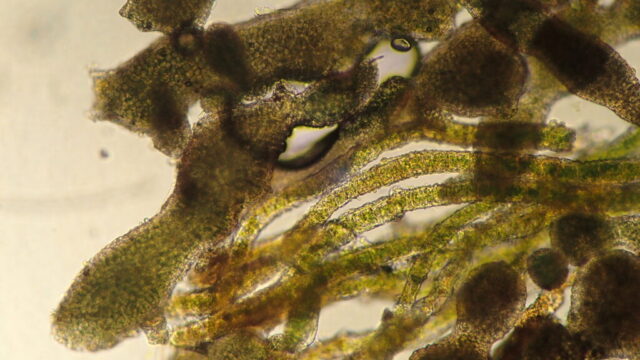
The cause of the lack of lichens is, I think, the quarry history and how it worked. From what I can make out online (so I may be wrong about this) it only ceased working in 1982 and basically involved the removal of the plentiful scree and cemented scree (what I would call “periglacial head”). The surfaces we see have only been exposed since the 1970s. Which explains why there aren’t many lichens on the limestone: there hasn’t been time for colonisation. The occasional stone with lichens on might well have fallen from above: we could hear rocks cracking and tumbling. The tiny columns of cemented scree that are left had some of the best bits of lichen, but these were often difficult to remove (let alone identify) pyrenocarps.
We spent some time looking at the hazels growing along the southern edge of the quarry. There’s the beginnings of a decent smooth bark community: Graphis species; Coniocarpon cinnabarinum and C. elegans; and probably some Eopyrenulas too (though I didn’t take any samples to check for the latter). But no Pertusarias yet- I think they take a little longer to arrive. With luck, they won’t get shaded out by other trees, brambles or ivy.
We called it a day in the middle of the afternoon, a bit earlier than I’d hoped. And our list was really rather short. Not that we hadn’t had a good time: there were lizards and a couple of slow worms on the way down the slab, the peregrines kept calling. I’d learnt something about limestone lichen ecology and succession. But I don’t think I’ll be stopping to look at the quarry again in the near future. Though maybe it’s time to go and have another look at the plateau.
Text and photos: Pete Martin